 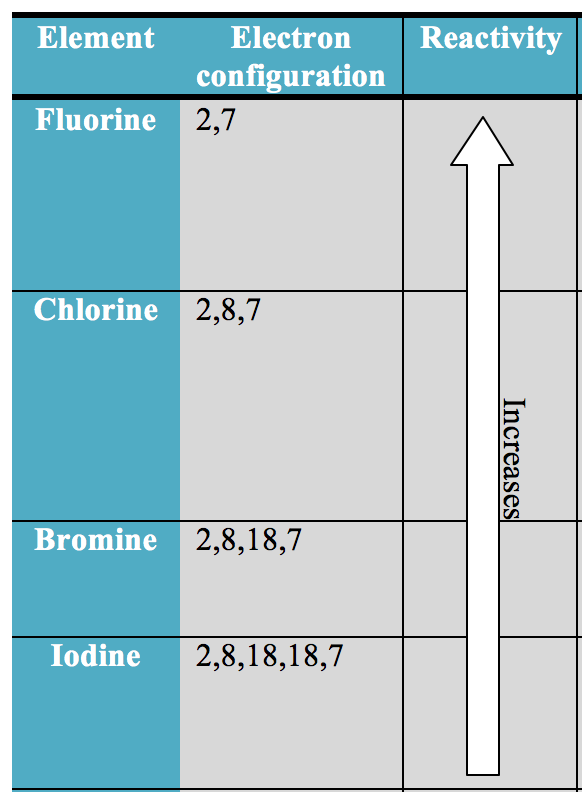
Livermorium isotopes 290Lv through 293Lv have been discovered the most stable livermorium isotope is 293Lv, which has a half-life of 0.061 seconds. In addition to the stable isotopes, some radioactive chalcogen isotopes occur in nature, either because they are decay products, such as 210Po, because they are primordial, such as 82Se, because of cosmic ray spallation, or via nuclear fission of uranium. Polonium has 42 isotopes, none of which are stable. Tellurium has eight stable or nearly stable isotopes, 31 unstable ones, and 17 isomers. Selenium has six observationally stable or nearly stable isotopes, 26 radioactive isotopes, and 9 isomers. Sulfur has four stable isotopes, 20 radioactive ones, and one isomer. Oxygen has three stable isotopes, and 14 unstable ones. Out of the six known chalcogens, one (oxygen) has an atomic number equal to a nuclear magic number, which means that their atomic nuclei tend to have increased stability towards radioactive decay. Density, melting and boiling points, and atomic and ionic radii tend to increase towards the chalcogens with higher atomic numbers. Electronegativity decreases towards the chalcogens with higher atomic numbers. All of the solid, stable chalcogens are soft and do not conduct heat well. Properties Atomic and physical Ĭhalcogens show similar patterns in electron configuration, especially in the outermost shells, where they all have the same number of valence electrons, resulting in similar trends in chemical behavior:Īll chalcogens have six valence electrons. 
Some of polonium's applications are due to its radioactivity. Tellurium compounds are mostly used in optical disks, electronic devices, and solar cells. Selenium's most common application is glassmaking. Sulfur is mostly converted into sulfuric acid, which is heavily used in the chemical industry. The primary use of elemental oxygen is in steelmaking. Livermorium has been synthesized in particle accelerators. Polonium is most available in naturally occurring actinide-containing materials. 
Selenium and tellurium are produced as byproducts of copper refining. Sulfur is extracted from oil and natural gas. Oxygen is generally obtained by separation of air into nitrogen and oxygen. This trend also occurs with chalcogen pnictides and compounds containing chalcogens and carbon group elements. Not counting oxygen, organic sulfur compounds are generally the most common, followed by organic selenium compounds and organic tellurium compounds. There are numerous organic chalcogen compounds. Sulfur has more than 20 allotropes, oxygen has nine, selenium has at least eight, polonium has two, and only one crystal structure of tellurium has so far been discovered. Tellurium often has unpleasant effects (although some organisms can use it), and polonium (especially the isotope polonium-210) is always harmful as a result of its radioactivity. Selenium is an important nutrient (among others as a building block of selenocysteine) but is also commonly toxic. All of the naturally occurring chalcogens have some role in biological functions, either as a nutrient or a toxin. Lighter chalcogens are typically nontoxic in their elemental form, and are often critical to life, while the heavier chalcogens are typically toxic. They have relatively low atomic radii, especially the lighter ones. Their most common oxidation states are −2, +2, +4, and +6. All of the chalcogens have six valence electrons, leaving them two electrons short of a full outer shell. Selenium, tellurium and polonium were discovered in the 19th century, and livermorium in 2000. Sulfur has been known since antiquity, and oxygen was recognized as an element in the 18th century. The word "chalcogen" is derived from a combination of the Greek word khalkόs ( χαλκός) principally meaning copper (the term was also used for bronze/ brass, any metal in the poetic sense, ore or coin), and the Latinized Greek word genēs, meaning born or produced. Often, oxygen is treated separately from the other chalcogens, sometimes even excluded from the scope of the term "chalcogen" altogether, due to its very different chemical behavior from sulfur, selenium, tellurium, and polonium. Group 16 consists of the elements oxygen (O), sulfur (S), selenium (Se), tellurium (Te), and the radioactive elements polonium (Po) and livermorium (Lv). 
This group is also known as the oxygen family. The chalcogens (ore forming) ( / ˈ k æ l k ə dʒ ə n z/ KAL-kə-jənz) are the chemical elements in group 16 of the periodic table. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
